
“Bzzzzz, bzzzzz, bzzzzz.” This sound circles the ear before the bite is felt. A female Anopheles mosquito rises from an open gutter at Olebu Zamzam in the Ga Central Municipality of the Greater Accra Region of Ghana. She is barely visible in the dusk, yet she carries the malaria parasite from one person to another.
Earlier in her life, she mated once in mid-air and stored enough sperm in a tiny organ called the spermatheca to fertilise eggs for the rest of her life.
Soon after, she searched for blood to help her eggs mature. She found a small pool of water trapped in waste, laid hundreds of eggs, and within days new mosquitoes emerged from the same polluted spot.
Pollution as a Breeding Ground
Across sub-Saharan Africa, this cycle repeats daily. According to the World Health Organisation, about 95 per cent of global malaria deaths occur in the African region. In 2024, an estimated 610,000 people died from the disease, most of them children under five.
Discarded plastics, cans, tyres, and blocked drains create ideal breeding sites for mosquitoes. Aedes aegypti thrives in such containers and transmits dengue and yellow fever, while polluted stagnant water supports Anopheles gambiae, the primary malaria vector in Africa.
Because only females bite and transmit disease, most control measures focus on stopping them through insecticide-treated bed nets, indoor residual spraying, larval source management, and improved housing.
Looking Beyond the Female
Far less attention is given to the male mosquito, yet new research is beginning to look at him as part of the solution. Specifically, the work of Target Malaria, in partnership with Imperial College London and the University of Oxford, explores gene drive technology that alters mosquito reproduction.
Dr Federica Bernardini, Research Associate in the Department of Life Sciences at Imperial College London sheds light on this approach.
“We know malaria is caused by a parasite called Plasmodium and that infection happens through the bite of an infected female mosquito. It is treatable and preventable, yet hundreds of thousands still die each year, mostly in Africa,” she says.
Her team studies the genes that influence how mosquitoes reproduce, feed, survive, and support parasite development.
“These features are genetically determined. If we disrupt certain genes, we can affect how mosquitoes multiply,” she explained.
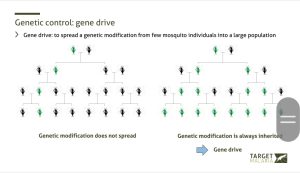
How Gene Drive Changes Inheritance
Gene drive ensures near-100% inheritance of the modification
Under normal inheritance, a genetic change passes to only half of the offspring.
“This is Mendelian inheritance. A modification is passed to about 50 per cent of the progeny, which is not enough to make a real difference in the wild unless very large numbers are released,” she said.
Gene drive technology alters this pattern.
“We ensure the modification is copied onto the matching gene inside the reproductive cells. Instead of 50 per cent inheritance, it becomes close to 100 per cent. All the offspring inherit the modification,” Dr Bernardini explained.
In laboratory cages, the effect is clear.
“As generations pass, the frequency of the modification increases while the number of eggs laid by females drops until they cannot produce eggs at all. The population collapses in the cage,” she said.
Targeted Science and Community Dialogue
The approach is precise. Only specific malaria vectors are targeted, including Anopheles gambiae, Anopheles coluzzii, Anopheles arabiensis, and Anopheles funestus.
“The strategy involves releasing genetically modified males in future field trials (subject to regulatory approval). When they mate with wild females, the females lose the ability to produce sperm. Over time, the mosquito population drops because it cannot support the next generation,” she said.
Environmental studies have examined the role of these mosquitoes in the ecosystem.
“Studies suggest they are not a keystone species in the food chain, and other mosquito species may occupy the same ecological niche without transmitting malaria,” she notes.
As the world marks World Malaria Day on April 25, she emphasises that science must move with public engagement.
“Our goal is to explain the science clearly and engage communities and decision makers before any field use is considered,” she said.
Beyond vector control, researchers at Imperial College London have also developed portable Lab-on-the-chip diagnostic tools such as Dragonfly and Lacewing, designed to provide rapid and accurate detection of infections, including malaria, from a small blood sample.
This report is part of the UK-Ghana ST&I Media Training Programme.
By Afia Agyapomaa Ofosu, science journalist
E-mail: prissyof@yahoo.com


